A) When a reaction produces a mixture of enantiomers, it is often very difficult to separate them. A mixture of products is often not useful.
B) Often stereoisomers of a particular compound will have very different biological effects on an organism. Only one isomer is biologically helpful, and the other may be harmful.
C) Often only one stereoisomer is biologically active, and coupling reactions are often used for the production of biological materials.
D) All of the choices are true.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the products of the following reaction. 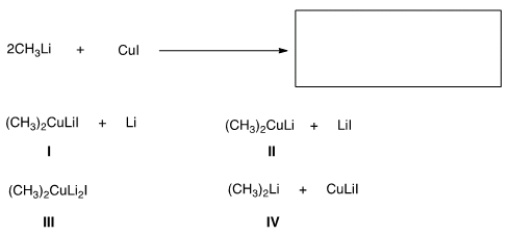
A) A
B) B
C) C
D) D
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name for the type of intermediate X formed in the reaction sequence below? 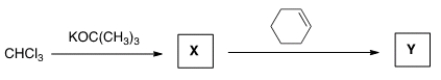
A) a carbocation
B) a carbanion
C) a free radical
D) a carbene
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reactant in the following reaction? 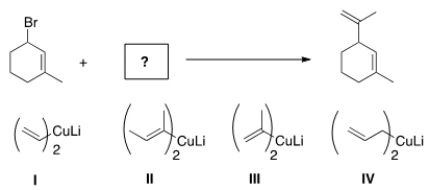
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the starting material that would be used to form the following product in a ring-closing metathesis reaction utilizing a Grubbs catalyst? 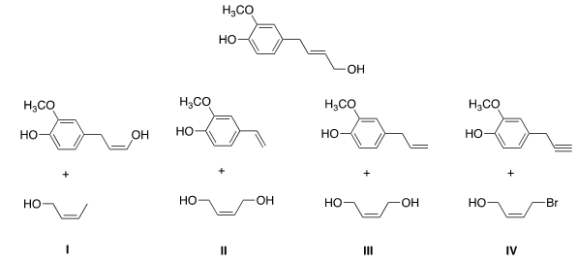
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the appropriate sequence of reagents that will produce the target molecule shown below from ethylbenzene? 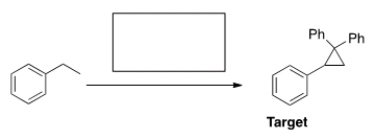
A) (1) HBr, H2O2; (2) KOC(CH3) 3 ; (3) MCPBA; (4) CH2=CPh2
B) (1) NBS, h ; (2) KOC(CH3) 3 ; (3) CHBr3, KO(CH3) 3 ; (4) a. LiCuPh2, b. H2O
C) (1) H2SO4(aq.) , ; (2) MCPBA; (3) CHBr3, KO(CH3) 3 ; (4) CH2=CPh2
D) (1) Br2, FeBr3 ; (2) MCPBA; (3) CHBr3, KO(CH3) 3 ; (4) Ph2COCl
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would be the major organic product of the following Heck reaction? 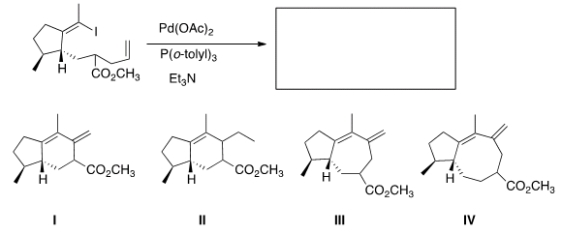
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is not true about the Simmons-Smith reaction?
A) The Simmons-Smith reaction involves the formation of a free carbene.
B) The Simmons-Smith reaction uses the reagents zinc-copper couple.
C) The Simmons-Smith reaction is stereospecific.
D) The Simmons-Smith reaction involves CH2I2 reacting with a copper-activated zinc reagent.
F) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Identify the products of the following reaction. 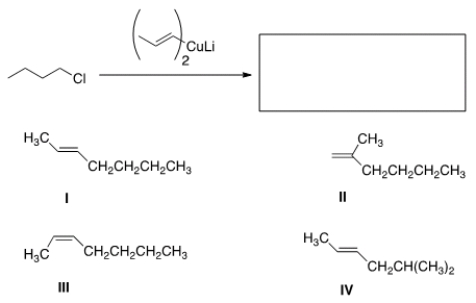
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the structure of the organic product Y formed in the following reaction sequence. 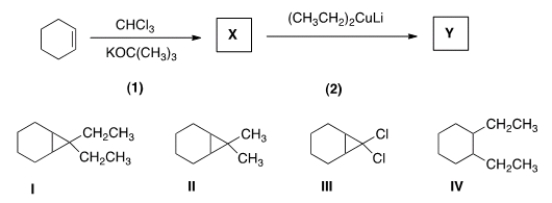
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 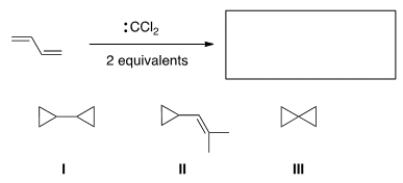
A) I
B) II
C) III
D) None of the choices is correct
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the major organic product of the following reaction. 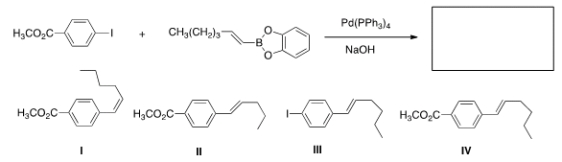
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a step in the Suzuki reaction?
A) Oxidative addition of R-X to the palladium catalyst
B) Substitution of the R group to the palladium catalyst
C) Transfer of the alkyl group from the organoborane to palladium
D) Reductive elimination of R-R, forming the new C-C bond
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 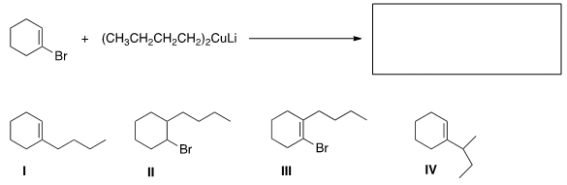
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Dichlorocarbene reacts with an alkene to form a cyclopropane derivative. In this reaction the dichlorocarbene acts as a(n)
A) Lewis base.
B) electrophile.
C) nucleophile.
D) Brønsted-Lowry base.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the reactions listed below is not stereospecific?
A) Suzuki reaction
B) organocuprate coupling reaction
C) Heck reaction
D) Simmons-Smith reaction
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the Grubbs catalyst synthetically important?
A) Because it only requires dilute concentrations of the reactants.
B) Because it produces only stereospecific products.
C) Because it produces only stereoselective products.
D) Because it provides a synthetic pathway for ring-closing metathesis reactions.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Compound X can be synthesized via a ring-closing metathesis reaction when treated with a Grubbs' catalyst. What is a possible structure of the starting material for this reaction? 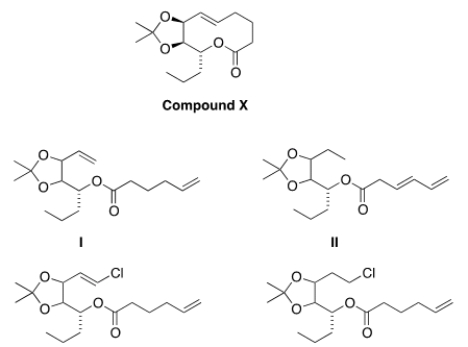
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What starting material could yield the following compound if Simmons-Smith conditions were used? 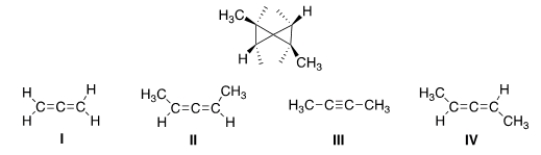
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly describes a reaction yielding a dichorocarbene?
A) The reaction of chloroform with KOC(CH3) 3.
B) The reaction of chloroform with Zn(Cu) .
C) The reaction of chloroform with (CH3) 2CuLi.
D) The reaction of diazomethane with KOC(CH3) 3.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 41
Related Exams