A) 4 K
B) 30 K
C) 70 K
D) 100 K
E) 130 K
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal in the simple cubic lattice will have ________________ atom(s) per unit cell.
A) 1
B) 2
C) 3
D) 4
E) 8
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
a.State the essential requirements for hydrogen bonding to be important in a compound. b.List four properties of water which are significantly influenced by the presence of hydrogen bonding.
Correct Answer

verified
a.The compound must contain hydrogen bon...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Draw a fully labeled phase diagram (P versus T)of a substance whose solid phase can melt due to applied pressure (i.e. ,solid is less dense than liquid).Clearly label the triple point and the critical temperature on your diagram.
Correct Answer

verified
 A is the triple poi...
A is the triple poi...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Liquid ammonia (boiling point = -33.4°C) can be used as a refrigerant and heat transfer fluid.How much energy is needed to heat 25.0 g of NH3(l) from -65.0°C to -12.0°C? 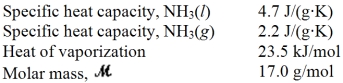
A) 5.5 kJ
B) 6.3 kJ
C) 39 kJ
D) 340 kJ
E) 590 kJ
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Of the five major types of crystalline solid,which would you expect each of the following to form? (e.g. ,H2O: molecular) a.Sn b.Si c.KCl d.Xe e.F2
Correct Answer

verified
a.metallic
b.network...View Answer
Show Answer
Correct Answer
verified
b.network...
View Answer
Multiple Choice
The energy needed to increase the surface area of a liquid is
A) capillary action.
B) surface tension.
C) viscosity.
D) cohesion.
E) specific elasticity.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal such as chromium in the face-centered cubic lattice will have ________________ atom(s) per unit cell.
A) 1
B) 2
C) 3
D) 4
E) 10
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances does not exist in the indicated solid type?
A) graphite - network
B) Na - metallic
C) SiO2 - molecular
D) NaCl - ionic
E) diamond - network
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonia's unusually high melting point is the result of
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonding.
E) ionic bonding.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy gap between the conduction band and the valence band is large for
A) conductors.
B) semiconductors.
C) superconductors.
D) insulators.
E) alloys.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Consider the phase diagram shown below.  a.What phase(s)is/are present at point A?
b.What phase(s)is/are present at point B?
c.Name point C and explain its significance.
d.Starting at D,if the pressure is lowered while the temperature remains constant,describe what will happen.
a.What phase(s)is/are present at point A?
b.What phase(s)is/are present at point B?
c.Name point C and explain its significance.
d.Starting at D,if the pressure is lowered while the temperature remains constant,describe what will happen.
Correct Answer

verified
a.liquid
b.solid,liquid and gas
c.C is t...View Answer
Show Answer
Correct Answer
verified
b.solid,liquid and gas
c.C is t...
View Answer
Multiple Choice
Examine the following phase diagram and identify the feature represented by point B. 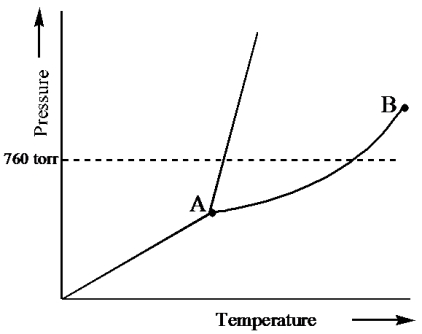
A) melting point
B) triple point
C) critical point
D) sublimation point
E) boiling point
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When liquid bromine is cooled to form a solid,which of the following types of solid would it form?
A) atomic
B) metallic
C) molecular
D) ionic
E) covalent network
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Strontium metal crystallizes in a cubic unit cell which has an edge length of 612 pm.If the mass of an atom of Sr is 87.62 amu,and the density of Sr metal is 2.54 g/cm3,calculate the number of atoms per unit cell.(1 amu = 1.661 10-24 g. )
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Polonium crystallizes in the simple cubic lattice.What is the coordination number for Po?
A) 3
B) 4
C) 6
D) 8
E) 12
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs is arranged with the particle of higher polarizability listed first?
A) CCl4,CI4
B) H2O,H2Se
C) C6H14,C4H10
D) NH3,NF3
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Use molecular orbital band diagrams to explain why metals are good conductors but semiconductors are not.
Correct Answer

verified
 In metals,the conduction and valence ba...
In metals,the conduction and valence ba...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following should have the highest surface tension at a given temperature?
A) CH4
B) CF4
C) CCl4
D) CBr4
E) CI4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Iron has a body-centered cubic unit cell,and a density of 7.87 g/cm3.Calculate the edge length of the unit cell,in pm.(The atomic mass of iron is 55.85 amu.Also,1 amu = 1.661 10-24g. )
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 89
Related Exams