A) Ionic bond
B) Nonpolar covalent bond
C) Polar covalent bond
D) Coordinate covalent bond
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be an ionic compound?
A) NCl3
B) BaCl2
C) CO
D) SO2
E) SF4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these polar covalent bonds would have the greatest percent ionic character?
A) H -Br
B) H - Cl
C) H - F
D) H- I
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond in which of the following pairs of atoms would be the least polar (i.e., lowest percent ionic character) ?
A) C - Cl
B) C - C
C) C - H
D) O - C
E) N - C
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis dot symbol for the S 2- ion is
A) (![]() )
)
B) (![]() 2-)
2-)
C) (S2-)
D) ( ![]() 2-)
2-)
E) (![]() )
)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the atoms listed below is the most electronegative?
A) Li
B) Cs
C) P
D) As
E) Ge
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formal charge on the sulfur atom in the resonance structure of sulfur dioxide which has one single bond and one double bond is
A) 0
B) +1
C) -1
D) +2
E) -2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of lone pairs in NCl3 is
A) 6
B) 8
C) 9
D) 10
E) 13
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use bond energies to estimate the enthalpy change for the reaction of one mole ofCH4 with chlorine gas to give CH3Cl and hydrogen chloride. BE(C-H) = 414 kJ/mol BE(C-Cl) = 326 kJ/mol BE(H-Cl) = 432 kJ/mol BE(Cl-Cl) = 243 kJ/mol
A) -101 kJ/mol
B) -106 kJ/mol
C) +331 kJ/mol
D) +106 kJ/mol
E) +101 kJ/mol
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assuming the octet rule is obeyed, how many covalent bonds will a neon atom form to give a formal charge of zero?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Each of the three resonance structures of NO3- has how many lone pairs of electrons?
A) 7
B) 8
C) 9
D) 10
E) 13
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Write a Lewis structure for the nitrate ion, NO3-, showing all non-zero formal charges.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be a covalent compound?
A) Rb2O
B) BaO
C) SrO
D) SeO2
E) MnO2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The covalent bond with the greatest polarity would form in which of the atom pairs below?
A) Br - Br
B) S - O
C) C - P
D) C - O
E) B - O
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solids would have the highest melting point?
A) NaI
B) NaF
C) MgO
D) MgCl2
E) KF
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Write the Lewis structure for the product that forms when boron trifluoride combines with ammonia.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the best Lewis structure for the fulminate ion, CNO-, what is the formal charge on the central nitrogen atom?
A) +2
B) +1
C) 0
D) -1
E) -2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Write a Lewis structure for the phosphate ion, PO43-, that expands the octet to minimize formal charge and if necessary places negative formal charges on the most electronegative atom(s).
Correct Answer

verified
Correct Answer
verified
Short Answer
Use the Born-Haber cycle to calculate the lattice energy of NaBr(s)given the following data: H(sublimation)Na = 109 kJ/mol I1 (Na)= 496 kJ/mol Bond energy (Br-Br)= 192 kJ/mol EA (Br)= 324 kJ/mol Hf (NaBr(s))= -361 kJ/mol
Correct Answer

verified
Correct Answer
verified
Short Answer
Consider the hypothetical element Y with electron-dot formula 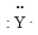 Give the formula for the simplest compound this element forms with chlorine.
Give the formula for the simplest compound this element forms with chlorine.
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 118
Related Exams