A) 2.2 × 10-3/s
B) 0.70 mmHg/s
C) 2.2 × 10-3/mmHg·s
D) 6.7 × 10-6/mmHg·s
E) 5.2 × 10-5/mmHg·s
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Substitute natural gas can be synthesized by passing carbon monoxide and hydrogen over Ni or Co at 400°C. 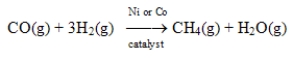 This process is an example of homogeneous catalysis.
This process is an example of homogeneous catalysis.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An increase in the temperature of the reactants causes an increase in the rate of reaction.The best explanation for this behavior is that as the temperature increases,
A) the concentration of reactants increases.
B) the activation energy decreases.
C) the collision frequency increases.
D) the fraction of collisions with total kinetic energy greater than Ea increases.
E) the activation energy increases.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The rate determining step must be the first step of a reaction mechanism in all cases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction A + 2B products has been found to have the rate law, rate = k[A] [B]2.While holding the concentration of A constant, the concentration of B is increased from x to 3x.Predict by what factor the rate of reaction increases.
A) 3
B) 6
C) 9
D) 27
E) 30
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The gas phase reaction of nitrogen dioxide and carbon monoxide was found experimentally to be second-order with respect to NO2, and zero-order with respect to CO below 25°C. NO2 + CO NO + CO2 Which one of the following mechanisms is consistent with the observed reaction order?
A) NO2 + 2CO ![]() N + 2CO2 fast N + NO2 2NO slow
N + 2CO2 fast N + NO2 2NO slow
B) NO2 + 2CO N + 2CO2 slow N + NO2 2NO fast
C) NO2 + NO2 NO3 + NO fast NO3 + CO NO2 + CO2 slow
D) NO2 + NO2 NO3 + NO slow NO3 + CO NO2 + CO2 fast
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In general, to calculate the activation energy for an elementary step given the rate constants at two different temperatures, which equation should be used?
A) ln([A]t/[A]o) = - kt
B) t1/2 = ln2/k
C) ln(k1/k2) = Ea/R( (T1 - T2) /T1T2)
D) 1/[A]t = kt + 1/[A]o
E) Rate = k[A]
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
It is possible for the following overall reaction to consist of a one step mechanism: 2A products
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction A + 2B products has the rate law, rate = k[A][B]3.If the concentration of B is doubled while that of A is unchanged, by what factor will the rate of reaction increase?
A) 2
B) 4
C) 6
D) 8
E) 9
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When acetaldehyde at a pressure of 364 mmHg is introduced into an evacuated 500.mL flask at 518°C, the half-life for the second-order decomposition process, CH3CHO CH4 + CO, is 410.s.What will the total pressure in the flask be after 1.00 hour?
A) 327 mmHg
B) 654 mmHg
C) 37 mmHg
D) 691 mmHg
E) 728 mmHg
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction C4H10 C2H6 + C2H4 has an activation energy (Ea) of 350 kJ/mol, and the Ea of the reverse reaction is 260 kJ/mol.Estimate H, in kJ/mol, for the reaction as written above.
A) -90 kJ/mol
B) +90 kJ/mol
C) 350 kJ/mol
D) -610 kJ/mol
E) +610 kJ/mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the overall chemical reaction shown below, which one of the following statements can be rightly assumed? 2H2S(g) + O2(g) \9\rarr\) 2S(s) + 2H2O(l)
A) The reaction is third-order overall.
B) The reaction is second-order overall.
C) The rate law is, rate = k[H2S]2 [O2].
D) The rate law is, rate = k[H2S] [O2].
E) The rate law cannot be determined from the information given.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At 700 K, the rate constant for the following reaction is 6.2 × 10-4 min-1. 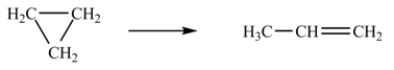 How many minutes are required for 20% of a sample of cyclopropane to isomerize to propene?
How many minutes are required for 20% of a sample of cyclopropane to isomerize to propene?
A) 1,120 min
B) 360 min
C) 3710 min
D) 1.4 × 10-4 min
E) 280 min
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitrous oxide (N2O) decomposes at 600°C according to the balanced equation 2N2O(g) 2N2(g) + O2(g) A reaction mechanism involving three steps is shown below.Identify all of the catalysts in the following mechanism. Cl2(g) 2Cl(g) N2O(g) + Cl(g) N2(g) + ClO(g) (occurs twice) ClO(g) + ClO(g) Cl2(g) + O2(g)
A) Cl
B) Cl2
C) ClO
D) N2O
E) ClO and Cl
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction BrO3- + 5Br-+ 6H+ 3Br2 + 3H2O at a particular time, - [BrO3-]/ t = 1.5 × 10-2 M/s.What is - [Br-]/ t at the same instant?
A) 13 M/s
B) 7.5 × 10-2 M/s
C) 1.5 × 10-2 M/s
D) 3.0 × 10-3 M/s
E) 330 M/s
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
A nuclear stress test utilizes a gamma-emitting radioisotope such as thallium-201 to follow the flow of blood through the heart - first at rest, and then under stress.The first-order rate constant for the decay of thallium-201 is 9.5 x 10-3 hr-1.Calculate how long it takes for the amount of thallium-201 to fall to 5.0% of its original value.
Correct Answer

verified
Correct Answer
verified
Short Answer
The rate constant for the first-order decomposition of C4H8 at 500°C is 9.2 × 10-3 s-1.How long will it take for 10.0% of a 0.100 M sample of C4H8 to decompose at 500°C?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A reaction was experimentally determined to follow the rate law, Rate = k[A] where k = 0.15 s-1.Starting with [A]o = 0.225M, how many seconds will it take for [A]t = 0.0350M
A) 3.4 x 10-2 s
B) 5.3 x 10-3 s
C) 12 s
D) 160 s
E) 1.3 s
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The first-order decomposition, A products, has a rate constant of 0.150 s-1.Starting with [A]o = 0.350 M, how much time is required for [A]t = 0.125 M?
A) 6.86 s
B) 2.98 s
C) 34 s
D) 1.50 s
E) 4.62 s
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
At a certain temperature, the data below were collected for the reaction below.
2ICl + H2 I2 + 2HCl.
Determine the rate constant for the reaction. 
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 130
Related Exams