A) 0.039 M-1·s−1
B) 6.9 M-1·s−1
C) 0.50 M-1·s−1
D) 0.54 M-1·s−1
E) 25 M-1·s−1
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Carbon dating may be used to date (once living)materials that are between 100 and 40,000 years old.The half-life of the first-order decay of carbon-14 is 5730 years.What percentage of carbon-14 remains in a sample after 40,000 years.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following steps must occur for an enzyme to catalyze a reaction?
A) The reactant,often called the substrate,must bind to the enzyme.
B) The chemical reaction must be halted.
C) The products of the reaction must attach to the enzyme.
D) The quantity of the enzymes in the reaction should keep increasing.
E) The reactant must bind to the product rather than the enzyme.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The average rate of disappearance of ozone in the following reaction is found to be 7.01 × 10-3 atm/s. 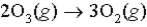 What is the rate of appearance of
What is the rate of appearance of 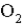 during this interval?
during this interval?
A) 4.67 × 10-3 atm/s
B) 7.01 × 10-3 atm/s
C) 16.4 × 10-3 atm/s
D) 172 × 10-3 atm/s
E) 10.5 × 10-3 atm/s
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a chemical reaction,the activation energy for the forward reaction is +181 kJ and the activation energy for the backward reaction is +62 kJ.What is the net energy change for the forward reaction?
A) -119 kJ
B) -62 kJ
C) +119 kJ
D) +181 kJ
E) +243 kJ
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The pre-exponential,A,in the Arrhenius equation is called the ________ factor.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the half-life of the first-order reaction if it takes 4.4 × 10-2 seconds for a concentration to decrease from 0.50 M to 0.20 M?
A) 2.5 × 10-2 s
B) 3.3 × 10-2 s
C) 1.6 s
D) 21 s
E) 27 s
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A suggested mechanism for the decomposition of ozone is as follows: O3 ![A suggested mechanism for the decomposition of ozone is as follows: O<sub>3</sub> O<sub>2</sub> + O fast equilibrium O + O<sub>3</sub> 2O<sub>2</sub> slow step What is the rate law predicted by this mechanism? A) Rate = B) Rate = k<sub>2</sub>[O] [O<sub>3</sub>] C) Rate = D) Rate = E) Rate =](https://d2lvgg3v3hfg70.cloudfront.net/TB7480/11eac9a9_4bc8_111d_acc3_bb04b9e3f1e5_TB7480_11.jpg) O2 + O fast equilibrium
O + O3
O2 + O fast equilibrium
O + O3 ![A suggested mechanism for the decomposition of ozone is as follows: O<sub>3</sub> O<sub>2</sub> + O fast equilibrium O + O<sub>3</sub> 2O<sub>2</sub> slow step What is the rate law predicted by this mechanism? A) Rate = B) Rate = k<sub>2</sub>[O] [O<sub>3</sub>] C) Rate = D) Rate = E) Rate =](https://d2lvgg3v3hfg70.cloudfront.net/TB7480/11eac9a9_4bc8_382e_acc3_317ead3179a9_TB7480_11.jpg) 2O2 slow step
What is the rate law predicted by this mechanism?
2O2 slow step
What is the rate law predicted by this mechanism?
A) Rate = ![]()
B) Rate = k2[O] [O3]
C) Rate = ![]()
D) Rate = ![]()
E) Rate = ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of NO and O2 produces NO2. 2 NO(g) + O2(g) → 2 NO2(g) The reaction is second-order with respect to NO(g) and first-order with respect to O2(g) .At a given temperature,the rate constant,k,equals 4.7 × 102 M-2s-1.What is the rate of reaction when the initial concentrations of NO and O2 are 0.025 M and 0.015 M,respectively?
A) 2.6 × 10-3 M/s
B) 4.4 × 10-3 M/s
C) 0.18 M/s
D) 2.0 × 10-8 M/s
E) 3.8 × 102 M/s
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How are the exponents in a rate law determined?
A) They are equal to the inverse of the coefficients in the overall balanced chemical equation.
B) They are determined by experimentation.
C) They are equal to the coefficients in the overall balanced chemical equation.
D) They are equal to the reactant concentrations.
E) They are equal to the ln(2) divided by the rate constant.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the initial rate data for the decomposition reaction, A → B + C Determine the rate expression for the reaction. [A],M -Δ[A]/Δt M/s 0) 084 12) 4 × 10-6 0) 063 9) 3 × 10-6 0) 042 6) 2 × 10-6
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At a given temperature,a first-order reaction has a rate constant of 2.1 × 10-3 s-1.How long will it take for the reaction to be 25% complete?
A) 660 s
B) 2100 s
C) 1500 s
D) 59 s
E) 140 s
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 72 of 72
Related Exams